No products in the cart.
Prednisolone EP Impurity E
Product Description
CAT No.
ALN-P027006
CAS No.
95815-58-0
Mol. F.
C21H28O6
Mol. Wt.
376.4
Stock
Please Inquire
Product Overview
Technical Data
Reference
RFQ
Product Overview
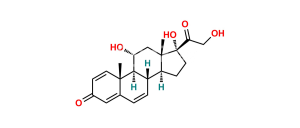
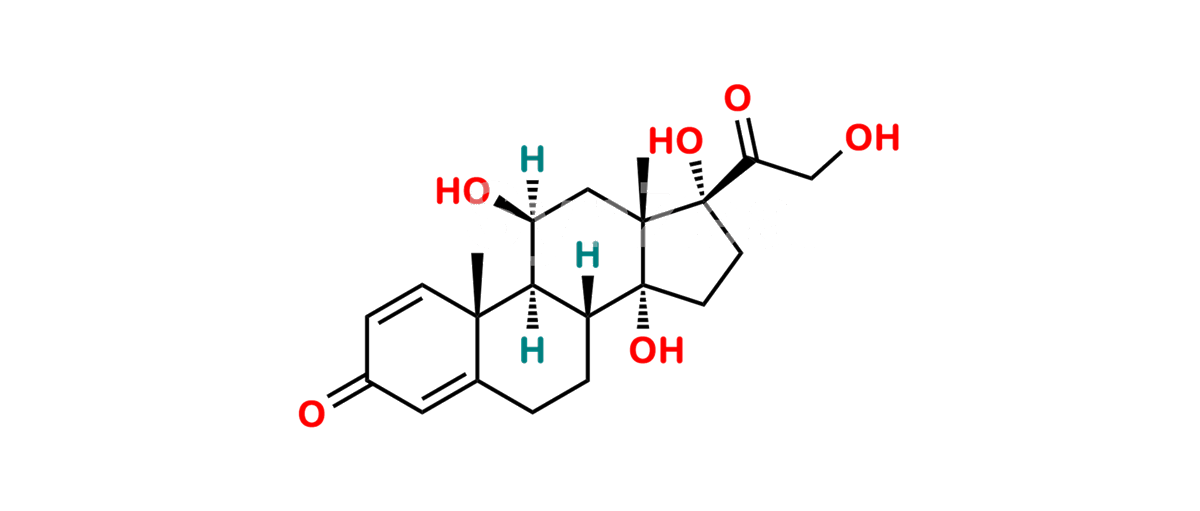
Chemical Name : 11β,14α,17,21-Tetrahydroxypregna-1,4-diene-3,20-dione (as per EP)
Smiles : O=C1C=C[C@]2(C)[C@@]3([H])[C@](O)([H])C[C@]4(C)[C@](C(CO)=O)(O)CC[C@@]4(O)[C@]3([H])CCC2=C1
Inchi : InChI=1S/C21H28O6/c1-19-5-3-11(23)7-14(19)15(24)8-12-13-4-6-21(27,17(26)10-22)20(13,2)9-16(25)18(12)19/h3,5,7,12-13,15-16,18,22,24-25,27H,4,6,8-10H2,1-2H3/t12-,13-,15+,16-,18+,19-,20-,21-/m0/s1
Synonym : 14α-Hydroxyprednisolone (EP)
Technical Data
Reference
Validated stability indicating RP-LC assay for determination of gatifloxacin and prednisolone acetatein ophthalmic preparations and biological samples
By Qadir, Muhammad A.; Shahzad, Shabnam; Ahmed, Mahmood; Razzaq, Syed S.; Shafiq, Muhmmad I.nFrom Latin American Journal of Pharmacy (2016), 35(5), 912-920
Novel stability indicating RP-HPLC method for the simultaneous estimation of moxifloxacin and prednisolone in bulk and their combined dosage form
By Potnuri, Naga Raju; Rao, G. Devala; Prasad, Y. Rajendra – From International Journal of Pharmaceutical Sciences and Research (2015), 6(5), 1965-1973
Development and validation of a new stability indicating reversed phase liquid chromatographic method for the determination of prednisolone acetate and impurities in an ophthalmic suspension
By Marley, Adrian; Stalcup, Apryll M.; Connolly, Damian – From Journal of Pharmaceutical and Biomedical Analysis (2015), 102, 261-266
RFQ