No products in the cart.
Abiraterone Impurity 5

Product Description
CAT No.
ALN-A006023
CAS No.
32138-69-5
Mol. F.
C19H27IO
Mol. Wt.
398.3
Stock
Please Inquire
Product Overview
Technical Data
Reference
RFQ
Product Overview
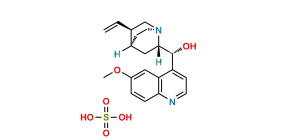
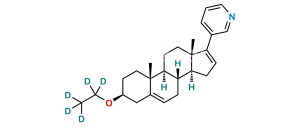
Chemical Name : (3S,8R,9S,10R,13S,14S)-17-Iodo-10,13-dimethyl-2,3,4,7,8,9,10,11,12,13,14,15-dodecahydro-1H-cyclopenta[a]phenanthren-3-ol
Smiles : O[C@H](C1)CC[C@@]2(C)C1=CC[C@]3([H])[C@]2([H])CC[C@@]4(C)[C@@]3([H])CC=C4I
Inchi : InChI=1S/C23H34O2/c1-5-16-7-9-20-19-8-6-17-14-18(25-15(2)24)10-12-23(17,4)21(19)11-13-22(16,20)3/h6-7,18-21H,5,8-14H2,1-4H3/t18-,19-,20-,21-,22+,23-/m0/s1
Synonym : 17-Iodoandrosta-5,16-diene-3β-ol
Technical Data
Reference
Identification, Characterization and HighPerformance Liquid Chromatography Quantification for Process-Related Impurities in Abiraterone Acetate Bulk Drug
Changjie Hu, Hanqiao Zhang, Menglin Zhang, Zhiyuan Mi, Jun Wang, Wenpin Lu, and Jiangtao Su*nJournal of Chromatographic Science, Volume 56, Issue 9, October 2018, Pages 802u2013811
stability indicating rp-hplc-pda method for determination of abiraterone acetate and characterization of its base catalyzed degradation product by lc-ms
– Journal of Chromatographic Science, Volume 56, Issue 9, October 2018, Pages 802–811
Department of Quality Assurance Techniques, maeer’S Maharashtra Institute of Pharmacy, MIT Campus, Kothrud, Pune 411038 – International Journal of Pharmacy and Pharmaceutical Sciences, Vol 8, Issue 2, 2016
RFQ