No products in the cart.
Daclatasvir Impurity 6

Product Description
CAT No.
ALN-D036028
CAS No.
74761-42-5
Mol. F.
C7H13NO4
Mol. Wt.
175.2
Stock
Please Inquire
Product Overview
Technical Data
Reference
RFQ
Product Overview
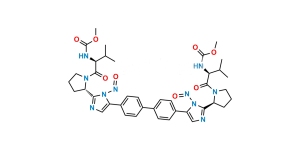
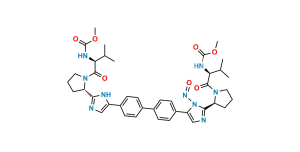
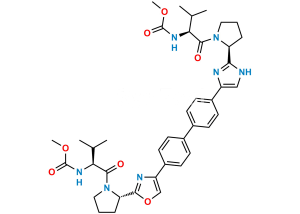
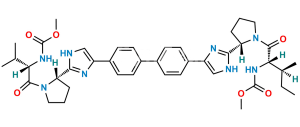
Chemical Name : (Methoxycarbonyl)-L-valine
Smiles : COC(N[C@@H](C(C)C)C(O)=O)=O
Inchi : InChI=1S/C26H28N6.H/c1-3-21(27-13-1)25-29-15-23(31-25)19-9-5-17(6-10-19)18-7-11-20(12-8-18)24-16-30-26(32-24)22-4-2-14-28-22;/h5-12,15-16,21-22,27-28H,1-4,13-14H2,(H,29,31)(H,30,32);/t21-,22-;/m0./s1/i;1+3
Technical Data
Reference
A validated stability-indicating reverse-phase high-performance liquid chromatography method for daclatasvir, identification and characterization of degradation products using LC-ESI-QTOF-MS
By Warghade, Snehal V.; Bothara, Kailash G.nFrom Asian Journal of Pharmaceutical and Clinical Research (2019), 12(5), 302-308
Development and validation of HPLC fluorescence and UPLC/DAD stability-indicating methods for determination of hepatitis C antiviral agent daclatasvir
By Kamal, Andra H.; Ismail, Nahla S.; Mabroijk, Mokhtar M.; Bebawy, Lories I.; Mekky, Mai A. – From Journal of AOAC International (2019), 102(4), 1125-1131
A stability-indicating UPLC method for the determination of potential impurities and its mass by a new QDa mass detector in daclatasvir drug used to treat hepatitis C infection
By Jagadabi, Varaprasad; Kumar, P. V. Nagendra; Mahesh, Kasthuri; Pamidi, Srinivasu; Ramaprasad, L. A.; Nagaraju, D. – From Journal of Chromatographic Science (2019), 57(1), 44-53
RFQ