No products in the cart.
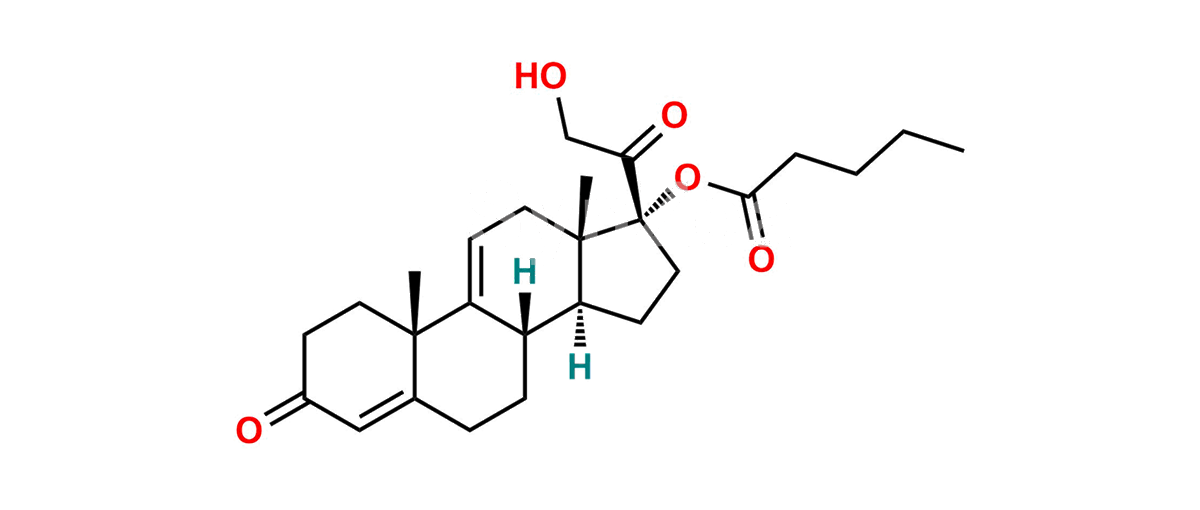
Δ9(11)-Hydrocortisone 17-Valerate
Product Description
CAT No.
ALN-H008051
CAS No.
NA
Mol. F.
C26H36O5
Mol. Wt.
428.6
Stock
Please Inquire
Product Overview
Technical Data
Reference
RFQ
Product Overview
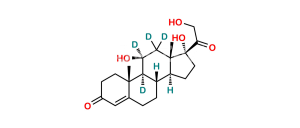
Chemical Name : (8S,10S,13S,14S,17R)-17-(2-Hydroxyacetyl)-10,13-dimethyl-3-oxo-2,3,6,7,8,10,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl pentanoate
Smiles : CCCCC(O[C@]1(C(CO)=O)CC[C@@]2([H])[C@]3([H])CCC4=CC(CC[C@]4(C)C3=CC[C@]12C)=O)=O
Inchi : InChI=1S/C23H31BrO6/c1-13(25)30-12-19(28)22(29)9-7-16-17-5-4-14-10-15(26)6-8-20(14,2)23(17,24)18(27)11-21(16,22)3/h10,16-18,27,29H,4-9,11-12H2,1-3H3/t16-,17-,18-,20-,21-,22-,23-/m0/s1
Technical Data
Reference
Comparative study of two different chromatographic approaches for quantitation of hydrocortisone acetate and pramoxine hydrochloride in presence of their impurities
Fawzia Ibrahim a , Asmaa Kamal El-Deen a,b , Kuniyoshi ShimizunJournal of Food and Drug Analysis Volume 26, Issue 3, July 2018, Pages 1160-1170
Novel stability indicating UHPLC method development and validation for simultaneous quantification of hydrocortisone acetate, pramoxine hydrochloride, potassium sorbate and sorbic acid in topical cream formulation
Lakshmi Narasimha RaoKatakamaThirupathiDongalabSanthosh KumarEttaboina – Talanta Open Volume 1, August 2020, 100004
Novel eco-friendly chromatographic determinations of hydrocortisone acetate, fusidic acid, their pharmacologically active impurities and pharmaceutical excipients: a comparative study
Maha M. Abdelrahman, Raghda Abdelmoneim Emam, Nouruddin W. Ali & Eglal A. Abdelaleem – Chemical Papers volume 74, pages2175–2187(2020)
RFQ