No products in the cart.
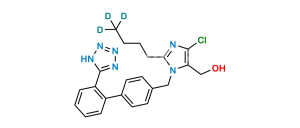
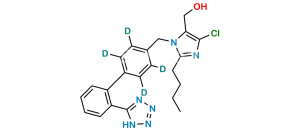
Losartan EP Impurity H

Product Description
CAT No.
ALN-L028009
CAS No.
133909-99-6
Mol. F.
C41H37ClN6O
Mol. Wt.
665.2
Stock
Please Inquire
Product Overview
Technical Data
Reference
RFQ
Product Overview
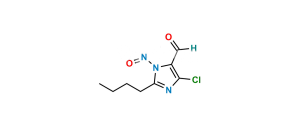
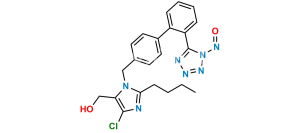
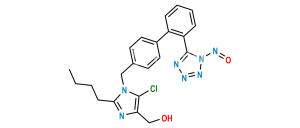
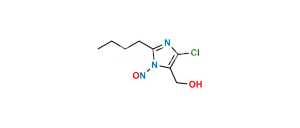
Chemical Name : 2-Butyl-4-chloro-1-[[2′-[2-(triphenylmethyl)-2H-tetrazol-5-yl][1,1′-biphenyl-4-yl]methyl]-1H-imidazole-5-methanol
Smiles : OCC1=C(Cl)N=C(CCCC)N1CC2=CC=C(C3=CC=CC=C3C4=NN(C(C5=CC=CC=C5)(C6=CC=CC=C6)C7=CC=CC=C7)N=N4)C=C2
Inchi : InChI=1S/C19H16O/c20-19(16-10-4-1-5-11-16,17-12-6-2-7-13-17)18-14-8-3-9-15-18/h1-15,20H
Synonym : N2-Trityl Losartan
Technical Data
Reference
Development and validation of a stability-indicating HPLC method for the simultaneous determination of Losartan potassium, hydrochlorothiazide, and their degradation products
Deanne L. Hertzog a,*, Jennifer Finnegan McCafferty b , Xueguang Fang b , R. Jeffrey Tyrrell c , Robert A. ReednJournal of Pharmaceutical and Biomedical Analysis 30 (2002) 747u2013760
An Efficient, Commercially Viable, and Safe Process for Preparation of Losartan Potassium, an Angiotensin II Receptor Antagonist
, Jennifer Finnegan McCafferty b , Xueguang Fang b , R. Jeffrey Tyrrell c , Robert A. Reed – Journal of Pharmaceutical and Biomedical Analysis 30 (2002) 747–760
Suri Babu Madasu,†,‡ N. A. Vekariya,
RFQ