No products in the cart.
Ondensatron Impurity 1
Product Description
CAT No.
ALN-O002010
CAS No.
68890-19-7
Mol. F.
C12H12BrNO
Mol. Wt.
266.1
Stock
Please Inquire
Product Overview
Technical Data
Reference
RFQ
Product Overview
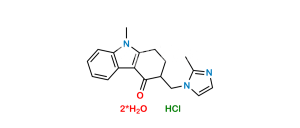
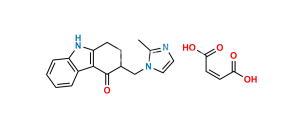
Chemical Name : 3-((2-Bromophenyl)amino)cyclohex-2-en-1-one
Smiles : O=C(CCC1)C=C1NC2=CC=CC=C2Br
Inchi : InChI=1S/C16H17N3O2/c1-9-8-17-14(10(2)15(9)21-4)16-18-12-6-5-11(20-3)7-13(12)19-16/h5-8H,1-4H3,(H,18,19)
Technical Data
Reference
Titania-based stationary phase in separation of ondansetron and its related compounds
Vu00e1clavu017diu017ekovsku00fd, RadimKuu010dera, Jiu0159u00edKlimeu0161, Jiu0159u00edDohnalnJournal of Chromatography A Volume 1189, Issues 1u20132, 2 May 2008, Pages 83-91
Degradation of ondansetron: Isolation and characterization impurity D ondansetron as A candidate reference standard impurity in drug
E. Kristiana, Asep Saefumillah, Emil Budianto – AIP Conference Proceedings 2242(1):040054
Identification, isolation and characterization of unknown impurity in Ondansetron Drug product
Hemant Madhusudan Gandhi, Nageswara Rao Gollapalli, Dr. Jaydeep Kumar D. Lilakar – International Journal of Advance Research, Ideas and Innovations in Technology (Volume 4, Issue 2) 1264-1271
RFQ