No products in the cart.
Pazopanib Impurity 13

Product Description
CAT No.
ALN-P013018
CAS No.
3934-20-1
Mol. F.
C4H2Cl2N2
Mol. Wt.
149
Stock
Please Inquire
Product Overview
Technical Data
Reference
RFQ
Product Overview
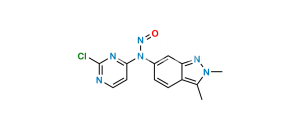
Chemical Name : 2,4-dichloropyrimidine
Smiles : ClC1=NC=CC(Cl)=N1
Inchi : InChI=1S/C21H23N7O2S.ClH/c1-13-5-6-15(11-19(13)31(22,29)30)24-20-9-10-23-21(25-20)27(3)16-7-8-17-14(2)28(4)26-18(17)12-16;/h5-12H,1-4H3,(H2,22,29,30)(H,23,24,25);1H
Technical Data
Reference
Analytical control of process impurities in Pazopanib hydrochloride by impurity fate mapping
Yan Li 1, David Q Liu, Shawn Yang, Ravinder Sudini, Michael A McGuire, Dharmesh S Bhanushali, Alireza S KordnJ Pharm Biomed Anal. 2010 Aug 1;52(4):493-507.
Determination and characterization of process impurities in pazopanib hydrochloride drug substance
musty sharada, ravichandra babu – Int J Pharm Pharm Sci, Vol 8, Issue 4, 97-102
Characterization of forced degradation products of pazopanib hydrochloride by UHPLC?Q?TOF/MS and in silico toxicity prediction
Prinesh N. Patel\xa0 Pradipbhai D. Kalariya\xa0 Mahesh Sharma\xa0 Prabha Garg\xa0 M. V. N Kumar Talluri\xa0 S. Gananadhamu\xa0 R. Srinivas – Journal of mass spectrometry Volume50, Issue7 July 2015 Pages 918-928
RFQ