No products in the cart.
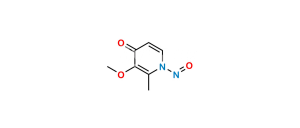
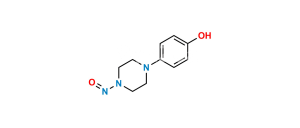
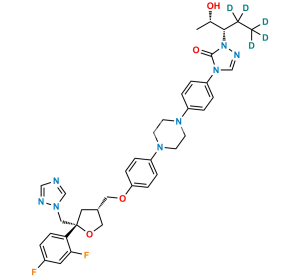
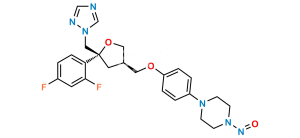
Posaconazole N,N-Dioxide Impurity

Product Description
CAT No.
ALN-P022070
CAS No.
1902957-95-2
Mol. F.
C37H42F2N8O6
Mol. Wt.
732.8
Stock
Please Inquire
Product Overview
Technical Data
Reference
RFQ
Product Overview
Chemical Name : 1-(4-(((3R,5R)-5-((1H-1,2,4-triazol-1-yl)methyl)-5-(2,4-difluorophenyl)tetrahydrofuran-3-yl)methoxy)phenyl)-4-(4-(1-((2S,3S)-2-hydroxypentan-3-yl)-5-oxo-1,5-dihydro-4H-1,2,4-triazol-4-yl)phenyl)piperazine 1,4-dioxide
Smiles : FC1=C(C=CC(F)=C1)[C@]2(C[C@H](COC3=CC=C([N](CC[N]4(C5=CC=C(N(C=NN6[C@@H](CC)[C@@H](O)C)C6=O)C=C5)=O)(CC4)=O)C=C3)CO2)CN7C=NC=N7
Inchi : InChI=1S/C37H42F2N8O3/c1-3-29(4-2)47-36(48)46(26-42-47)32-8-6-30(7-9-32)43-15-17-44(18-16-43)31-10-12-33(13-11-31)49-21-27-20-37(50-22-27,23-45-25-40-24-41-45)34-14-5-28(38)19-35(34)39/h5-14,19,24-27,29H,3-4,15-18,20-23H2,1-2H3/t27-,37+/m1/s1
Technical Data
Reference
Influence of cyclodextrin on posaconazole stability, release and activity: Improve the utility of the drug
By Santana, Ana Cristina Sousa Gramoza Vilarinho; Nadvorny, Daniela; da Rocha Passos, Taisa Daiany; de La Roca Soares, Monica Felts; Soares-Sobrinho, Jose LamartinenFrom Journal of Drug Delivery Science and Technology (2019), 53, 101153
Stability-indicating HPLC method development and structural elucidation of novel degradation products in posaconazole injection by LC-TOF/MS, LC-MS/MS and NMR
By Yang, Yidi; Zhu, Xi; Zhang, Fei; Li, Wei; Wu, Ying; Ding, Li – From Journal of Pharmaceutical and Biomedical Analysis (2016), 125, 165-177
Stability indicating HPLC method for the quantification of posaconazole and its related substances
By Govind, Sampath Kumar Reddy; Nagaraju, Ch. V. S.; Rajan, S. T.; Eshwaraiah, S.; Kishore, M.; Rakesh, M.; Chakravarthy, I. E. – From Pharma Chemica (2014), 6(1), 486-494, 9 pp..
RFQ